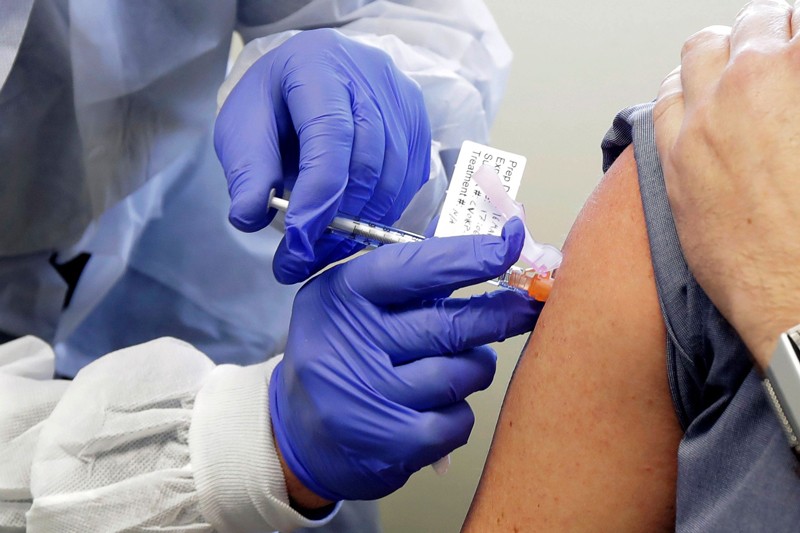
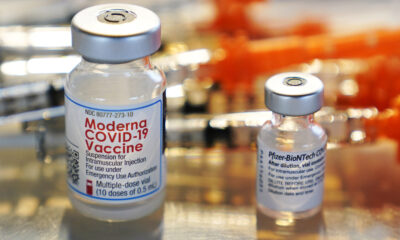
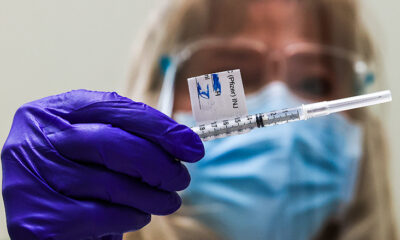
Johnson and Johnson said Wednesday it has started its stage three preliminary testing its potential Covid immunization.
J&J is the fourth drugmaker upheld by the Trump organization’s Covid-19 antibody program Operation Warp Speed to enter late-stage testing. The others are Moderna, Pfizer and AstraZeneca.
The preliminary will select up to 60,000 grown-up volunteers across 215 areas in the U.S. also, different nations, as per the National Institute of Allergy and Infectious Diseases. Members will be haphazardly chosen to get a portion of the possible antibody or a fake treatment, as per subtleties of the preliminary, which will decide if the immunization is sheltered and successful.
“Four COVID-19 vaccine candidates are in Phase 3 clinical testing in the United States just over eight months after SARS-CoV-2 was identified,” institute director Dr. Anthony Fauci said in a statement.
“This is an unprecedented feat for the scientific community made possible by decades of progress in vaccine technology and a coordinated, strategic approach across government, industry and academia,” he said. “It is likely that multiple COVID-19 vaccine regimens will be required to meet the global need. The Janssen candidate has showed promise in early-stage testing and may be especially useful in controlling the pandemic if shown to be protective after a single dose.”
J&J, whose Janssen auxiliary is situated in Belgium, has said it is utilizing similar advances it used to build up its trial Ebola antibody. It includes brushing hereditary material from the Covid with an altered adenovirus that is known to cause basic colds in people.
Preclinical examinations have indicated J&J’s potential Covid-19 antibody can create a promising reaction in nonhuman primates and hamsters.
Paul Stoffels, J&J’s boss logical official, told correspondents on a telephone call Tuesday that beginning phase preliminary information, which will be delivered “imminently,” shows the insusceptible reaction in people was “comparable to animals, which were protected.” He likewise said the antibody had all the earmarks of being all around endured, with manifestations, for example, fever settling inside 48 hours.
He said it will probably take a month and a half to two months to arrive at 60,000 members, permitting J&J to enlist a various companion. The preliminary will incorporate those both with and without comorbidities related with an expanded danger for movement to serious Covid-19, and will mean to enlist members in Argentina, Brazil, Chile, Colombia, Mexico, Peru, South Africa and the United States.
“There are a lot of what we call ‘readiness cohorts,’ where we have predetermined in high risk areas the specific populations we want to target,” he said. “The data teams have been working hard to find out where in the U.S., where in other parts of the world, we can get access to the people and parts of the population at high risk and diverse, which we need to have in the clinical trials.”
The advancement comes as President Donald Trump says the U.S. could locate a protected and successful immunization before the finish of October and have enough antibody portions to vaccinate each American by April.
Asked which drugmaker could be the first to win U.S. endorsement, Trump told “Fox News” on Monday that Pfizer, Moderna and AstraZeneca are advancing great. He included J&J’s immunization will “probably be a little later.”
The U.S. Branch of Health and Human Services reported on Aug. 5 that it arrived at an arrangement with Janssen, J&J’s drug auxiliary, worth around $1 billion for 100 million dosages of its antibody. The arrangement gives the national government the alternative to arrange an extra 200 million dosages, as per the declaration.
The stage three preliminary will be mutually financed by J&J, the National Institutes of Health and NIAID, Stoffels said Tuesday.
J&J has recently said it envisions conceivable immunization bunches for a potential crisis utilize approval accessible in mid 2021. The organization said it has proceeded with the scaling up of its assembling limit and stays on target to meet its objective of giving a billion portions of an antibody every year.
General wellbeing authorities and irresistible infection specialists state world pioneers will require a variety of medications and immunizations to crush the infection, which rose under 10 months prior.

 Technology4 weeks ago
Technology4 weeks ago
 Technology4 weeks ago
Technology4 weeks ago
 Technology4 weeks ago
Technology4 weeks ago
 Business4 weeks ago
Business4 weeks ago
 Technology4 weeks ago
Technology4 weeks ago
 Technology3 weeks ago
Technology3 weeks ago
 Technology3 weeks ago
Technology3 weeks ago
 Technology4 weeks ago
Technology4 weeks ago